Moritz Engelhardt, Dr. Kristin Grussmayer
Department of Bionanoscience, TU Delft, Netherlands
Background
The lab of Dr. Kristin Grussmayer develops light microscopy and analysis tools for life sciences and biophysics research, in particular addressing questions in molecular and cell biology. PhD student Moritz Engelhardt works within this group, with a project focused on studying protein aggregation in Huntington’s disease using cellular models, transfecting cells with plasmids containing the mutant Huntingtin protein and observing the effects of aggregation on cell health.
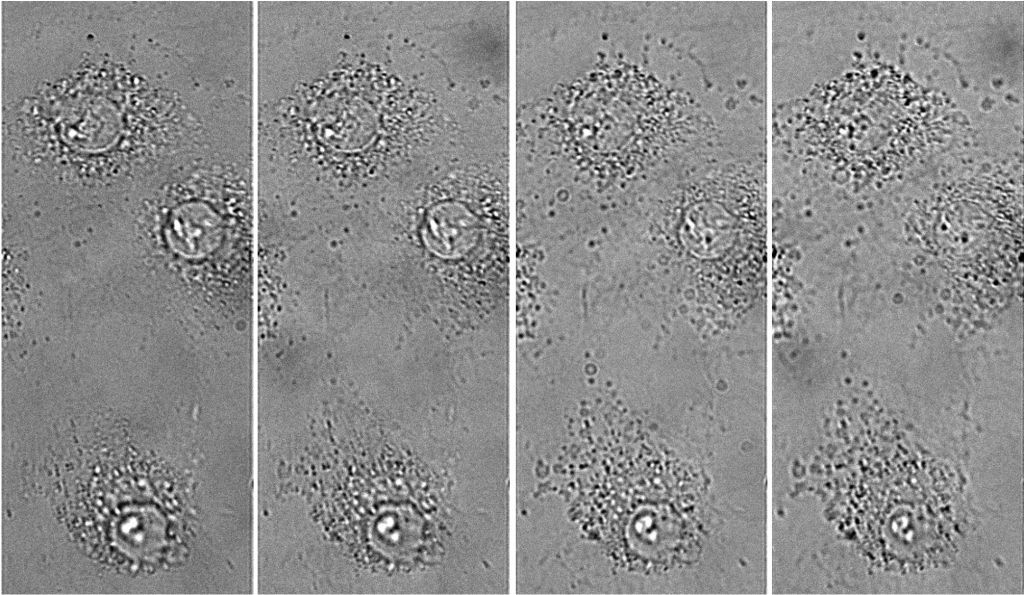
Moritz further explained his research, “We use a reconstruction algorithm that requires multiplane imaging to construct phase from brightfield images [1]. A prism splits light into different axial planes in the sample, and we can simultaneously look at different planes in the sample and use this to reconstruct the phase.”
“We also want to do subcellular segmentation in our phase images, we want to use a deep learning-based segmentation algorithm, to create the training data we use correlative fluorescence imaging. We take a phase image, then a fluorescence image to get the ground truth data of what part is the aggregate or organelle of interest.”
Challenge
While brightfield and phase imaging have a low light dose, there are plans for long-term live-cell fluorescence and super-resolution imaging (including SOFI), which will require more light.
For successful axial multiplane imaging and phase reconstruction, a prism is used to split the light. While the prism can restrict the field of view (FOV), Moritz and team place multiple of these FOVs across a single camera sensor for true simultaneous volumetric imaging and easier phase reconstruction. A camera with a large sensor is necessary in order to image multiple planes at once and capture fast dynamics.
Moritz outlined the other needs for a large sensor, “We can also use a large FOV when we go into fluorescence, where we can distribute axial information horizontally and chromatic channels vertically to facilitate high-speed multicolor volumetric imaging, all on a single big sensor.”
The large FOV of the Kinetix is great for collecting large amounts of information for our phase imaging and algorithm training, to capture dynamics at the millisecond scale in 3D.
Solution
The Kinetix sCMOS features one of the largest sensors of any scientific camera, the 29 mm diagonal FOV is easily able to contain FOVs from multiple planes and achieve simultaneously multiplane imaging. In addition, the Kinetix images at rapid speeds, achieving 500 fps across the entire sensor. When looking at multiple smaller FOVs, reducing the Kinetix imaging region further increases the speed and allows for the capture of truly rapid dynamics in live cellular samples.
Moritz shared his experience with using multiple Kinetix, “The Kinetix helps to not only look at static images but also time-resolved mitochondria dynamics, nucleocytosolic transport, and other fast samples. We can restrict our FOV and then go very fast to capture dynamics.”
“The big advantage of the Kinetix is the large sensor field of view for our phase imaging, and that we can be flexible for our future SMLM and SOFI experiments due to the high quantum, which is good. The Kinetix is great for collecting large amounts of information for our ground truth necessary for the algorithm training and to capture dynamics at the millisecond scale in 3D.”
Reference
[1] Descloux, A., Grußmayer, K.S., Bostan, E. et al. Combined multi-plane phase retrieval and super-resolution optical fluctuation imaging for 4D cell microscopy. Nature Photon 12, 165–172 (2018). https://doi.org/10.1038/s41566-018-0109-4

