Biophysics
Biophysics is a dynamic and growing field that applies physical methods and approaches to biological problems and systems. Biophysics shares an overlap with biochemistry, mathematics, mechanics, and computational sciences, making it a transdisciplinary and evolving field. Biophysical imaging contributes to drug discovery, learning more about the structure and function of biological molecules, and how cells and molecules interact.
When imaging in biophysics, researchers rely on highly sensitive and non-invasive analysis techniques. These techniques can be fluorescence-based, but also include light scattering, spectroscopy-based, and multi-photon techniques.

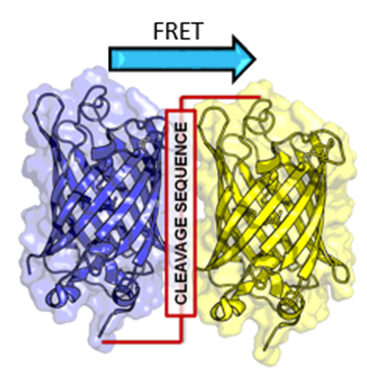
FRET
Similarly to how two magnets will only attract each other once close enough, energy can be transferred between fluorescently excited molecules if they are in extremely close proximity. This is the principle behind Förster Resonance Energy Transfer (FRET), which occurs when fluorophores are within 10 nm of each other, have overlapping spectra, and are in the correct orientation.
FRET can be used as a ‘spectroscopic ruler’ to measure the distance between molecules, to investigate binding dynamics, or to study protein conformational changes. FRET is an elegant technique with many uses and is widely used in biophysics.
FRAP
If intense light or long exposures are used with fluorophores, they can irreversibly lose their ability to emit light and are referred to as photobleached. While usually an issue in imaging, some techniques take advantage of this phenomenon, including fluorescence recovery after photobleaching (FRAP).
FRAP involves selectively photobleaching an area of a sample with intense laser light. Fluorescence in the bleached area will slowly recover as bleached fluorophores move out and healthy fluorophores from other areas move in. FRAP is useful in diffusion studies if targeted to the membrane or nucleus of live cells.

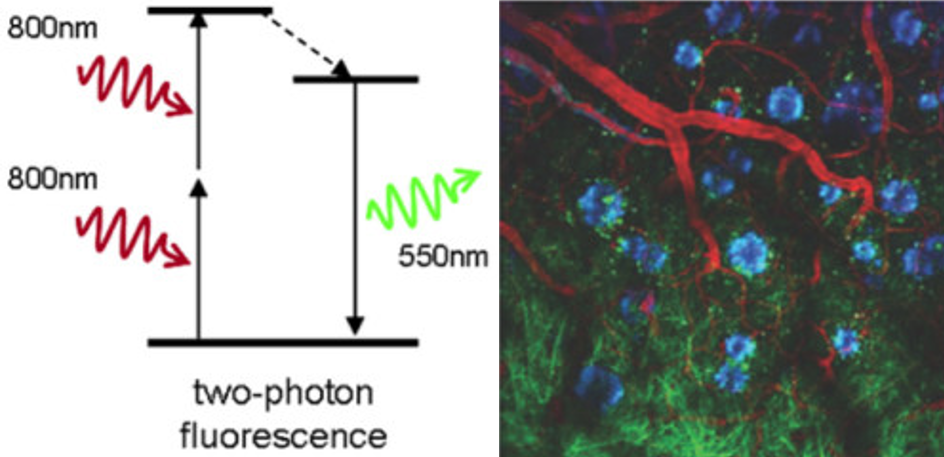
Two-Photon Microscopy
Conventional excitation in fluorescence microscopy typically involves one visible light photon. By using two lower-energy near-infrared (NIR) photons, standard fluorescent dyes can be excited in the NIR with localized excitation.
Two-photon microscopy allows for the imaging of very thick samples (up to several mm), live samples, and stable fluorescence over increased image depths. Two-photon microscopy can be used to image deep within live animals or whole brains, making it a very useful technique for certain situations.
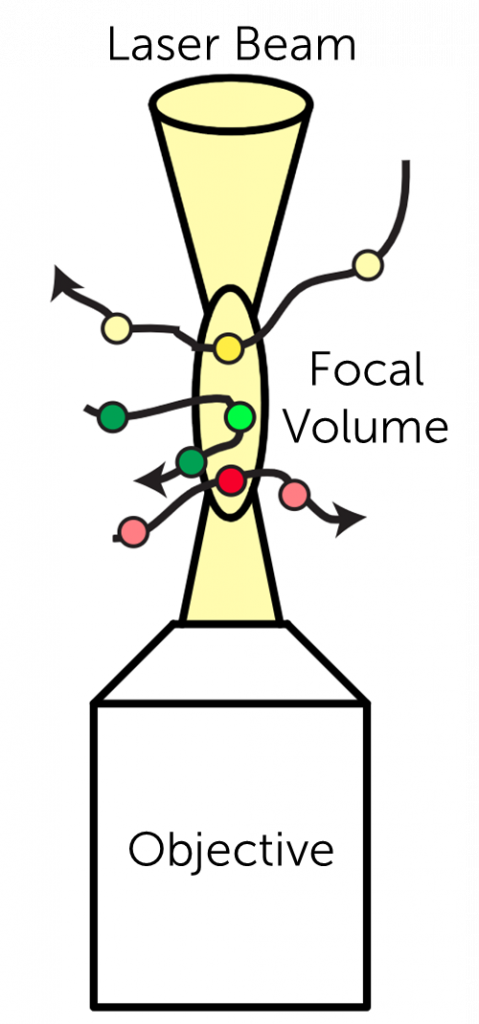
Fluorescence Correlation Spectroscopy
Fluorescence correlation spectroscopy (FCS) measures fluctuations in fluorescence intensity from fluorophores of interest, which can be caused by various dynamics, including the Brownian motion of particles.
FCS analysis provides fluorophore size and concentration, the viscosity of the solution, and the diffusion coefficient. FCS is an extremely sensitive analytical tool as it is able to measure an incredibly small amount of molecules (nano/picomolar concentrations) within a small volume.
Brillouin Microscopy
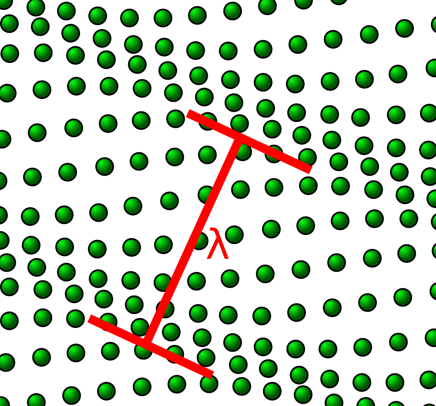
Brillouin microscopy is a powerful tool in the field of mechanobiology, using light scattering in order to measure the viscoelastic properties of 3D cells and tissues.
Brillouin is label-free, contract-free, and non-destructive, making use of the Brillouin light scattering that occurs when light interacts with material, such as a cell membrane or cytosol.
iSCAT
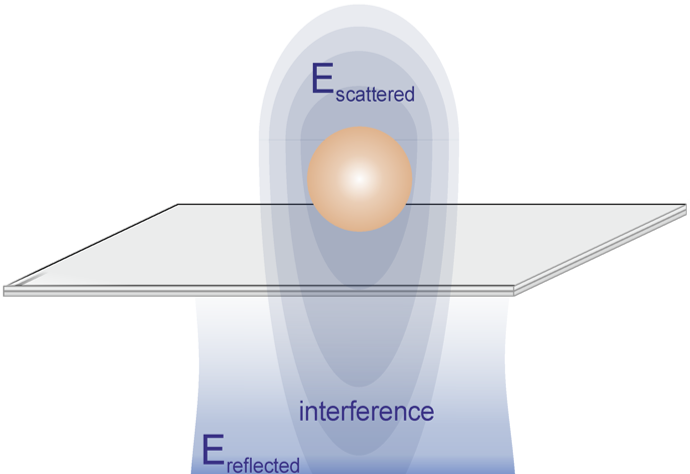
Interferometric scattering (iSCAT) allows for fast, high-resolution non-fluorescence detection of nanoscale samples such as viruses, DNA, and proteins.
iSCAT relies upon the interference of scattered light with a reference light field, allowing for tracking of molecules with extremely low signals.
Mesolens
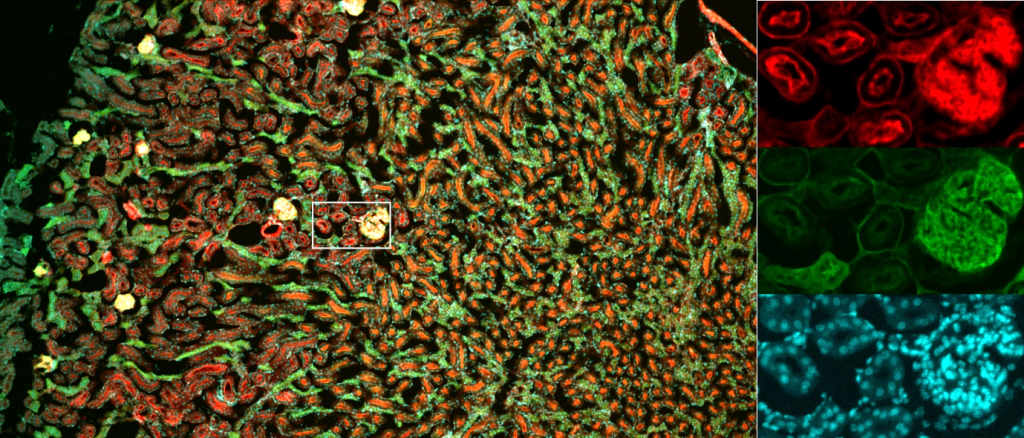
As sCMOS camera field of view (FOV) continues to grow larger, optics have developed in order to optimally match these big FOVs while retaining high levels of detail at low magnification.
The Mesolens is a giant custom-made microscope objective that allows for a huge 6 mm x 6 mm FOV, enabling the imaging of large tissue samples and whole live organisms at subcellular resolutions.
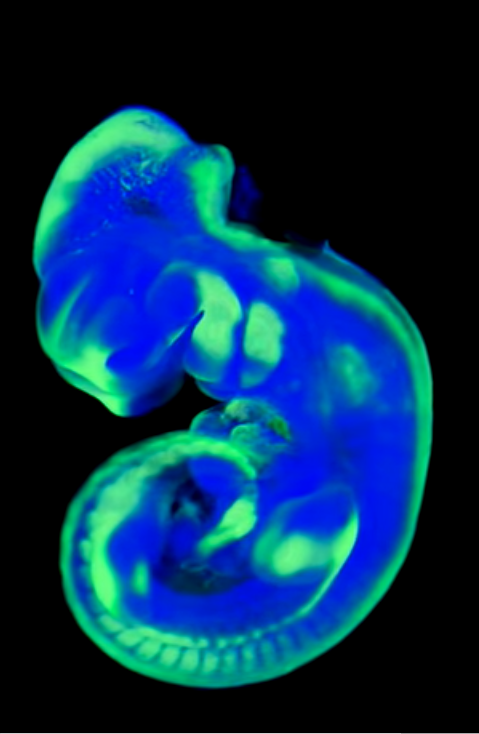
Fluorescence OPT
Optical Projection Tomography (OPT) works differently to other 3D optical imaging techniques, which typically rely on sectioning and section tomography. Projection tomography allows for imaging of larger 3D volumetric samples.
Images are taken from many angles as a large 3D sample (such as a whole mouse embryo) is rotated, building up a large high-resolution 3D image from back-projection algorithms.