Introduction
High content imaging (HCI) is a technique that benefits from highly specialized hardware and software. HCI can involve imaging many millions of cells, labeling with multiple fluorescent markers, and studying a large number of parameters. This results in thousands of interactions of different variables, meaning that the imaging system for HCI needs to be the first step in establishing a quantitative HCI setup. But should you buy, or build? This article will cover both options and the necessary features of a successful HCI system.
Important Features
When building or buying, it is important to know the most important features necessary in a HCI system. In January 2016, HTStec did a market survey on HCI and HCI systems to document the practices and preferences in the world of HCI, with the most important features highlighted in Fig.1.
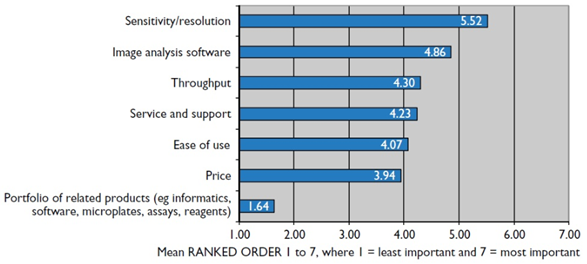
The most important feature was determined to be sensitivity and resolution, followed by analysis software and then throughput. The sensitivity and resolution of the system can be determined by the camera, objectives, magnification, light level, sample, and more. In terms of using a scientific camera, an sCMOS with a high quantum efficiency and a large field of view (FOV) would benefit researchers so that each image can contain as much content as possible. Imaging at high magnifications with high numerical aperture (NA) objectives will increase the image resolution, at the cost of FOV, so this compromise should be considered for a HCI system. Lower magnifications will allow larger areas to be imaged but at a lower resolution, but this may be necessary if large samples are being imaged. All factors in an imaging system affect the sensitivity and resolution, so a number of decisions need to be made when acquiring such a system.
Buying A HCI System
Buying a HCI system has the advantage of the all-in-one box, containing the light source, microscope, objectives, and sCMOS cameras. Broadly, these commercial HCI systems are either widefield, spinning disk confocal, or laser scanning cytometers. These instruments can have useful features such as multiple light sources (for flexible excitation), lasers and/or LEDs, confocal imaging systems, a range of magnifications (high and low NA), and associated custom software. These benchtop systems present a flexible solution for a variety of HCI experiments.
However, these systems can be prohibitively expensive, limiting researchers through cost. In order to perform HCI with a system while on a budget, it is often better to build your own HCI system, which also offers greater flexibility and the ability to customize the system to suit your specific imaging needs.
Building A HCI System
In theory, any instrument that produces a multi-parameter analysis of sample phenotypes (morphology, size, fluorescence channels) via imaging can be termed a HCI system. However, a number of conventional HCI systems consist of an automated brightfield/fluorescent microscope combined with image and data analysis software. It is also important to include high-speed data connections, a computer with powerful processors, and extensive data storage that is necessary to sort through the large datasets produced by HCI experimentation.
As HCI systems are often high-throughput, speed and FOV can become the limiting factors of the system. If a microscope limits the FOV, it also limits the data output of the system compared to a larger FOV imaging system. This means that any HCI system needs to be as fast as possible, while also taking in as much information as possible per image. An efficient HCI imaging system could involve:
- High camera speeds (hundreds/thousands of frames per second, only possible with low exposure)
- Low exposure times (to increase camera speed, also leads to a requirement for bright signal)
- Bright fluorescent signal (from optimized staining or high-intensity light sources)
- Low magnification (the lower the magnification, the larger the FOV)
- Large camera FOV (the bigger the camera sensor, the larger the FOV
- High numerical aperture (Increases microscope resolution)
- Less fluorescent channels (minimum of two fluorescent channels to recognize essential cell phenotypes)
- Discriminatory positive/negative controls (to streamline experimentation)
- Rapid autofocus (in order to image many different cell conditions)
- Precise moving stage (in order to XY scan across large samples/well plates reproducibly)
- Environmental controls (control of temperature and CO2/O2 for live cell culture and imaging)
- Ease of analysis for large datasets (slow analysis would bottleneck system)
The goal is to maximize data output and experimental efficiency, to detect and quantify critical features from hundreds/thousands of samples in a particular condition, as fast as possible.
However, it should be noted that a HCI system might be more limited in configuration than a research microscope, as it is likely to have fewer options when it comes to light sources, objectives, or cameras.
Light Sources
In most systems, the choices for light sources are a lamp, laser, or light-emitting diode (LED):
- Lamps are unsuitable due to their short useable lifetime, broad excitation wavelengths (including UV and IR), excess heat emission, fluctuation on the second timescale, and loss of intensity over the week/month timescale.
- Conventional lasers provide a fixed monochromatic excitation wavelength but often operate at substantial power levels and can be expensive. White light/tunable lasers are available, these are not commonly used on commercial HCI systems and may not yet be optimized.
- LEDs offer a stable light output over a long lifetime, allowing for greater consistency across images
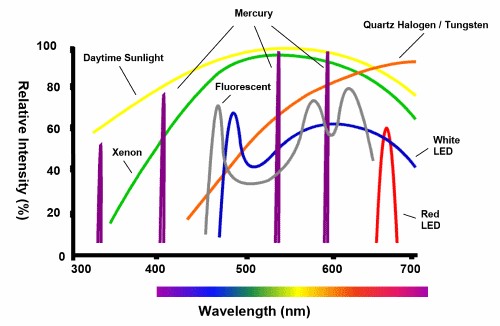
The light source for a HCI system should be bright with highly specific spectra, a long-life, not produce large amounts of heat, and reduce noise as much as possible from image to image. Most HCI systems would benefit from either lasers or LEDs depending on applications, with various intensities and wavelengths seen in Fig.2.
Objectives
While HCI can be done at any magnification, dependant on the sample, HCI can benefit from lower magnification objectives as these maximize the FOV and result in more data per image. However, these lower magnification objectives will have a lower NA and a lower resultant resolution. If detection is more important than sub-cellular resolution, the drop in resolving ability of the imaging system may be counteracted by the increase in the amount of data per image.
In addition, objectives with higher NAs will collect more light, potentially allowing for reduced exposure times, increased imaging speed, and increased throughout. The NA can be increased by using objectives with a different refractive index, namely water or oil immersion objectives. However, dealing with water or oil lenses can add challenges to automation, and are difficult to use with certain sample mounting methods, meaning that some HCI systems use air lenses with the highest NAs.
Cameras
HCI systems benefit most from scientific CMOS cameras as detectors. One limitation for HCI systems can be imaging speed, meaning that high speed, large FOV cameras can benefit HCI systems most. Other advantageous camera features include a large dynamic range (>20,000:1), broad spectral sensitivity (high quantum efficiency from 400-900 nm), and high resolution (>2000×2000 pixels). Cameras that can achieve each of these factors would be suited to HCI systems, but can produce large image files and would need to be paired with a powerful processing PC with large storage.
References
Buchser W, Collins M, Garyantes T, et al. Assay Development Guidelines for Image-Based High Content Screening, High Content Analysis and High Content Imaging. 2012 Oct 1 [Updated 2014 Sep 22]. In: Sittampalam GS, Grossman A, Brimacombe K, et al., editors. Assay Guidance Manual [Internet]. Bethesda (MD): Eli Lilly & Company and the National Center for Advancing Translational Sciences; 2004-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK100913/
