Introduction
Any microscope technique where the entire sample is exposed to light is known as ‘widefield’ imaging. The counterpart to widefield is confocal, where pinholes are used to block most of the light to and from the sample. This article will discuss widefield imaging and the most widely-used widefield techniques in microscopy.
Widefield Microscopes
In widefield, the entire sample is lit by a lamp light source from either below (an upright microscope) or from above (an inverted microscope). Upright microscopes are frequently used with fixed samples such as cells or tissues that have been treated and mounted onto microscope slides, whereas inverted microscopes are often better for imaging a sample immersed in liquid, as it would typically sink to the bottom and be easier to see from below with them microscope objectives. This allows for the imaging of suspension cells, as cells studied in the life sciences are typically either adherent (grow attached to a surface) or grow in suspension (cells suspended in fluid). Examples of an upright and inverted microscope can be seen in Fig.1.
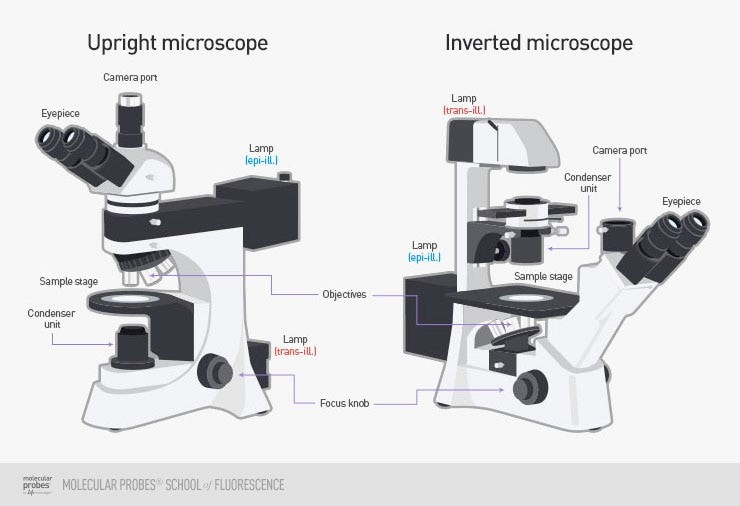
Widefield microscopes typically use a white light source (such as a lamp) is sufficient with some filters for fluorescence work. This also makes imaging simpler and image file sizes smaller, making it easier to work with widefield for applications such as cell documentation.
Widefield Techniques
Examples of widefield microscopy techniques are brightfield, differential interference contrast (DIC), phase contrast, and widefield fluorescence.
Brightfield microscopy is an accessible form of microscopy, where an entire sample is illuminated by a bright light. This approach involves little sample preparation and can be used to quickly and easily check on live cells or to produce supplementary data. However, using a contrast agent is highly recommended as most cell samples are transparent and will be challenging to resolve without a stain or dye. Cells are mostly water and when imaged on clear glass or plastic, it can be difficult to pick out smaller structures without some additional contrast.
For differential interference contrast (DIC), the sample is illuminated by light split into two polarized beams of light, when these beams recombine the differences in phase shift appear as contrast in the final image. Similarly to phase contrast, this technique isn’t suited to thicker samples and requires more of a technical setup than other techniques.
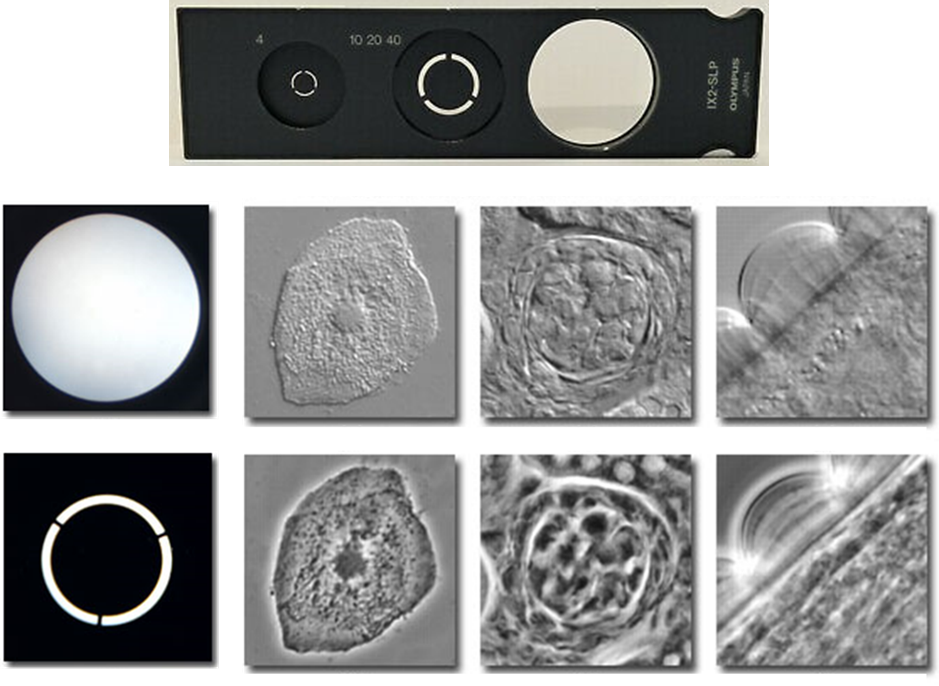
Phase-contrast microscopy provides better contrast than brightfield by using scattered light from the sample. By illuminating the sample with a ring of light and having another ring in front of the microscope viewfinder, parts of the sample that scatter light differently appear as darker or lighter on the image, giving it more contrast than standard brightfield microscopy. This improved contrast isn’t seen on thicker samples as it produces artifacts but works well with cell cultures. An example of these rings can be seen in Fig.2.
Widefield fluorescence microscopy is similar to brightfield but specific wavelengths of light are used in order to excite fluorescent molecules that the sample has been pre-treated with (although some samples are naturally auto-fluorescent). Samples can be stained with fluorescent markers for specific proteins or cell components, and then the fluorescence emission light from these markers forms an image. The fluorescence signal means that there is better contrast compared to other techniques, as by using specific wavelengths of light only the fluorescent molecules are emitting light, as opposed to the entire image being lit up. However, as the whole sample is lit using this light, the fluorescence signals from outside the viewing area can cause background fluorescence and blurry images.
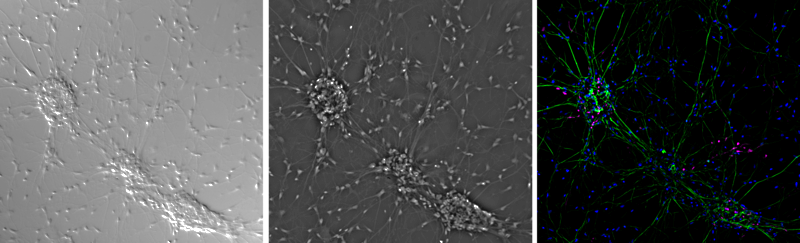
Out-Of-Focus Light
The main drawback is that as the whole sample is illuminated, while the focal plane receives light and can generate an image, the planes above and below the focal plane also receive light, resulting in out-of-focus light causing image degradation. Especially for fluorescence excitation, the resolution of a widefield system is limited due to the background fluorescence also being captured by the camera and decreasing the signal to noise ratio.
Certain widefield techniques avoid this problem, such as structured illumination microscopy (SIM), which uses patterns of light in order to generate a complex pattern, imaging based on the pattern interference allows for super-resolution levels of detail, resolving objects as small as 200 nm. More on SIM can be read on the SIM app note on our website.
Summary
Widefield imaging is the foundation of most cellular studies, allowing researchers to quickly and easily image samples with low levels of sample preparation or technical expertise needed. From brightfield to fluorescence imaging, widefield is a powerful and varied technique that many researchers are familiar with. While the technique may lack in resolution when compared to confocal or other advanced microscopy applications, widefield imaging has a firm place in research and will continue to develop over time.
