Prof. Bo Huang
Departments of Pharmaceutical Chemistry, Biochemistry and Biophysics; University of California, San Francisco (UCSF)
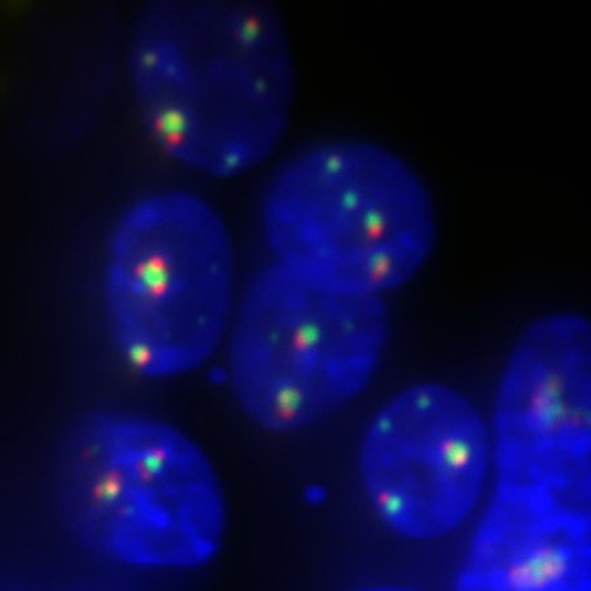
Background
Prof. Huang’s lab at UCSF aspires to generate a map of the entire proteome of endogenous proteins in human cells. Towards that goal they use and develop novel imaging modalities including eSPIM, a high numerical aperture epi-illumination form of selective plane illumination microscopy (SPIM). Using a galvanometer mirror to drive the position of an oblique light sheet, and a unique combination of re-imaging objectives angled to match the angle of the illumination sheet, they can image 34 slices per volume of 35x35x7 µm and collect 14 volumes per second. This allows for high-throughput volumetric imaging.
Challenge
Imaging with short exposure times is necessary to collect more than 450 frames per second and accurately combine them into 14 volumes, this demands the most out of the camera. Live-cell imaging at these rates will not allow increases of excitation laser power, as phototoxicity becomes an issue. Cameras with a limited field of view decrease the number of cells measured, requiring repeating experiments to generate sufficiently robust measurements. Prof. Huang told us they needed the camera to be most efficient at gathering signal, as well as have a high frame rate and appropriate pixel size for sampling the high resolution available in their optical design.
The combination of sensitivity, speed, field of view size and robust camera engineering of the Prime BSI has been very beneficial to our large-scale live cell imaging experiments.
Solution
Prof. Huang told us they found the 95% quantum efficiency, low pattern/read noise, and 6.5 µm pixel of the Prime BSI gave them the light-gathering efficiency and sampling size to meet their needs. The angled light-sheet only fills half the sensor area allowing up to 125 frames per second. Prof Huang told us they found that the Prime BSI was substantially more robust than another, previously tested, camera from a different vendor that used the same sensor.

