Prof. Dr. Helge Ewers
Institute Of Chemistry and Biochemistry, Freie Universität Berlin, Germany
Background
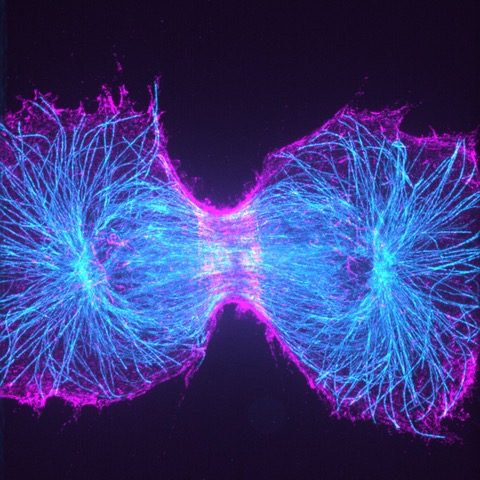
The laboratory of Prof. Ewers moves in a number of different research directions, one of these deals with the septin cytoskeleton. Septins are a family of essential, conserved GTP-binding proteins that form heteromeric, non-polar complexes that further assemble into filamentous structures. Septins have conserved functions in cell division and form part of the contractile actomyosin structure at the cleavage furrow in dividing cells. Currently, the Ewers laboratory uses super-resolution microscopy methods to understand the nanoscopic organization of septin structures in cell division.
Challenge
One technique used to understand the septin organization in the Ewers laboratory is the new Expansion microscopy technique. Expansion microscopy achieves higher resolution by the physical expansion of the sample rather than optically. When the target molecules are spaced wider than in the native state, regular microscopes can yield a much higher resolution. Expanding the sample into both X- and Y-directions however means that imaging the same cell will take more space on a camera chip. This is a significant challenge if a cell is expanded fourfold into X- and Y-directions, which means a 16-fold increase in area that must be imaged.
“Without the large chip [of the Prime BSI] we could not image the entire cell, and without the matched pixel size, we could not realize the gain in resolution through the expansion process. The results are stunning large images of septin structures in our genome-edited cells.”
Solution
Fortunately, the Teledyne Photometrics Prime BSI camera has a very large field of view. The Ewers laboratory uses this camera on their custom-made spinning disc microscope, where it does not only provide a large field of view, but the 6.5 μm pixel size enables true sub-resolution imaging with expansion microscopy at about the same sensitivity of an EMCCD camera. “Without the large chip,” Dr. Ewers said, “we could not image the entire cell. And without the matched pixel size, we could not realize the gain in resolution through the expansion process. The results are stunning large images of septin structures in our genome-edited cells.”

