Dr. Stephan Daetwyler, Prof. Reto Fiolka
Fiolka Lab, UT Southwestern Medical Center, Dallas, TX
Background
The lab of Prof. Reto Fiolka develops new, transformative technologies to image across scales: from sub-cellular imaging to imaging of whole organs. In the Fiolka Lab, Dr. Stephan Daetwyler is a postdoctoral researcher who builds, programs and applies advanced light-sheet microscopy systems to image dynamic processes in live biological organisms.
Dr. Daetwyler told us more of his work, “Amongst other innovations, the Fiolka lab has been a pioneer in a technique known as axially swept light-sheet microscopy or ASLM. ASLM excels in high-resolution 3D imaging of subcellular structures and signaling over extended volumes. I apply ASLM to study the dynamic behavior of single cells in developing zebrafish embryos. Interestingly, the development of vasculature is a highly dynamic process, and many thin, fine sprouts are formed. To reveal the behavior of these subcellular structures in vivo, ASLM is ideal.”
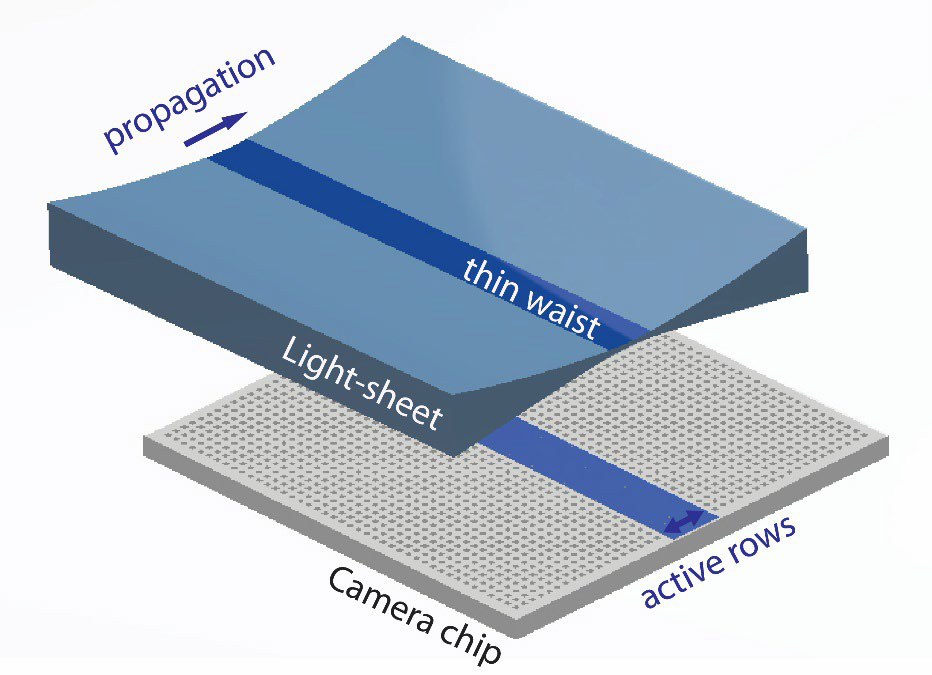
Challenge
ASLM requires a fast and flexible scientific camera to best synchronize the camera readout with the sweep of the light sheet. Therefore, light-sheet techniques such as ASLM require advanced camera modes that can control the camera readout speed and direction at a high timing accuracy.
Dr. Daetwyler discussed this need, “In ASLM we need to sweep this narrowly-focused beam across the camera chip and co-ordinate acquisition with the sweep of the light sheet.”
In addition, due to sweeping light sheet illumination, parts of the sample are only illuminated for a short time compared to conventional light-sheet acquisition. This results in comparably low levels of signal, meaning a sensitive camera with a high quantum efficiency is required.
I’m very happy with the Prime BSI Express, it’s a reliable camera that pairs well with our imaging techniques.

Solution
The Prime BSI Express is a powerful, flexible solution for 3D ASLM imaging. It allows for sub-cellular resolutions across a large sample. Furthermore, the presence of a 16-bit HDR mode with a low-noise CMS mode allows for high sensitivity imaging at a high dynamic range.
Importantly, the Prime BSI Express also features Programmable Scan Mode (PSM), where the direction and speed of camera readout can be controlled. This is ideal for techniques such as ASLM where the beam sweep had to be synchronized with camera readout.
Dr. Daetwyler told us of his experience with the Prime BSI Express, “I developed imaging software for the Prime BSI Express and Programmable Scan Mode using Python and PyVCAM. The setup was as easy as it can get for scientific cameras, the Prime BSI Express is a reliable camera that does what I want it to do!”

