Spinning Disk Confocal Microscopy
Confocal microscopy addresses two significant challenges in biological imaging that conventional fluorescence microscopy cannot overcome. Firstly, biological specimens are 3-dimensional structures so to fully understand them we often need to construct 3-dimensional images. Secondly, many processes biologists would want to study occur inside biological structures, but other cell features can block a clear view.
Confocal microscopy uses optical sectioning to take multiple, thin, 2-dimensional slices of a sample to construct a 3-dimensional model from them. This is made possible through the addition of a pinhole into the same focal plane as the sample to block out-of-focus light. Spinning disk confocal microscopy increases the speed of this technique by using multiple pinholes etched into an opaque disk which, when spun, scans the pinholes across the entire image.
Spinning disk confocal microscopy is, essentially, a light rejection technique so one of the main challenges is to collect as many of the emitted photons as possible so light intensity can be reduced to lessen the impact of photobleaching and photodamage on samples, which means that high camera sensitivity is of critical importance.

Prime 95B
Extremely sensitive, 95% quantum efficient sCMOS camera with 11 µm pixels and EMCCD level detection.
Go beyond EMCCD for spinning disk confocal microscopy with the back-illuminated Prime 95B, which features an equivalent level of detection but with a faster speed, larger field of view and no EM-gain aging or excess noise.
The Prime 95B allows exposure times to be lowered significantly to increase acquisition speed and reduce photobleaching and photodamage to the lowest levels possible on an sCMOS camera.
The results of a comparison between the Prime 95B and EMCCD for spinning disk confocal microscopy can be seen below.
AlexaFlour488-Phalloidin labelled actin cytoskeletal structure was imaged using spinning disk confocal microscopy.
The signal-to-noise improvement of the Prime 95B can be clearly seen. The Prime 95B image looks cleaner, details are sharper and structural elements are identified that can’t be seen with the EMCCD camera.

Prime BSI
High sensitivity, 95% quantum efficient, sCMOS camera with 6.5 µm pixels and 1.0 e– read noise.
The high quantum efficiency and low read noise combined with the balanced 6.5 µm pixel size offers high sensitivity imaging whilst achieving Nyquist sampling with the most popular objective magnifications used for spinning disk confocal microscopy.
The Prime BSI offers an alternative to the Prime 95B when resolution is more important than extreme sensitivity.

Kinetix
High sensitivity, 95% quantum efficient sCMOS camera with an incredibly high 400 fps full-frame speed and a massive 29.4 mm diagonal field of view.
The speed of the Kinetix significantly outperforms typical sCMOS devices. With a full-frame framerate of 400 fps and a 10 megapixel sensor, the Kinetix delivers over 4000 megapixels/second.
The high quantum efficiency and low read noise combined with the balanced 6.5 µm pixel size also delivers the sensitivity needed to get the highest image quality from a spinning disk system without sacrificing resolution.

Customer Stories
Spinning Disk Confocal
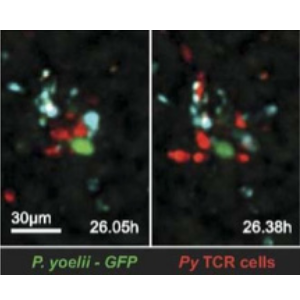
“Due to phototoxicity and speed of imaging, the Evolve 512 EMCCD camera is essentially running at single-molecule sensitivities in our experiments. We conducted side-by-side camera comparisons… Only the Evolve was suitable for this research.”
Super-Resolution Spinning Disk Confocal
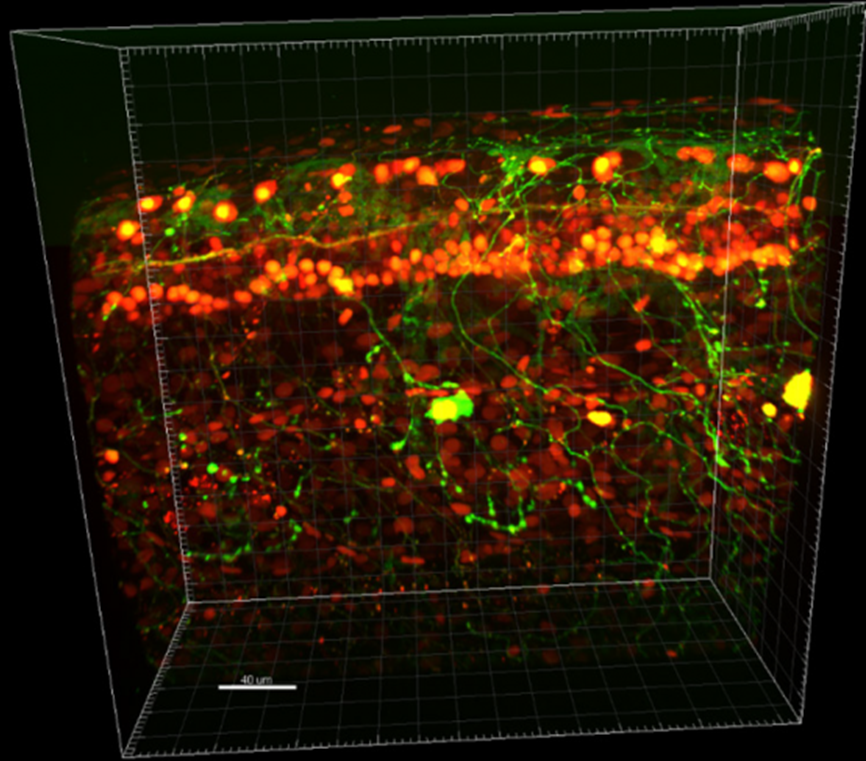
“The system combines the high speed of the spinning disk with the field of view and sensitivity of the Photometrics cameras. Together it allows us to do live imaging with weak samples, giving great performance.”
Live Cell Spinning Disk Microscopy
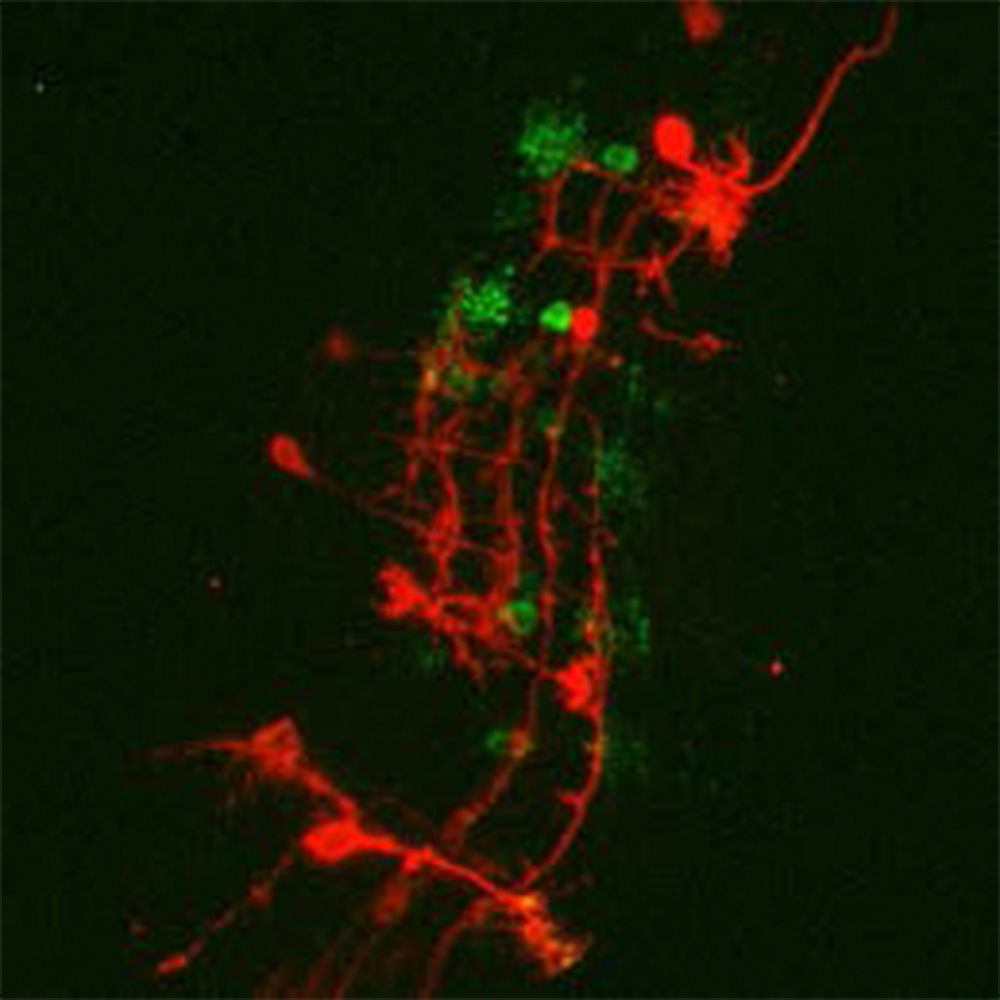
“Photometrics cameras are reliable with great performance. We can now visualize things that we could not see before [with the Prime 95B].”



